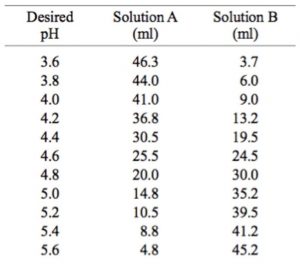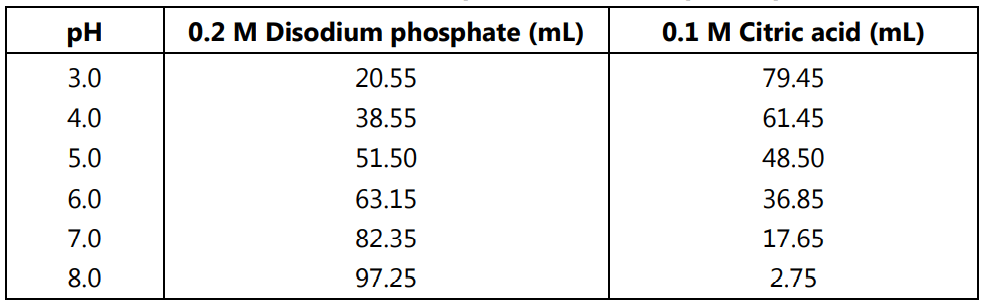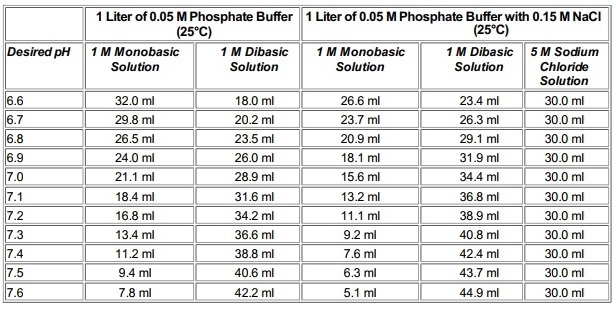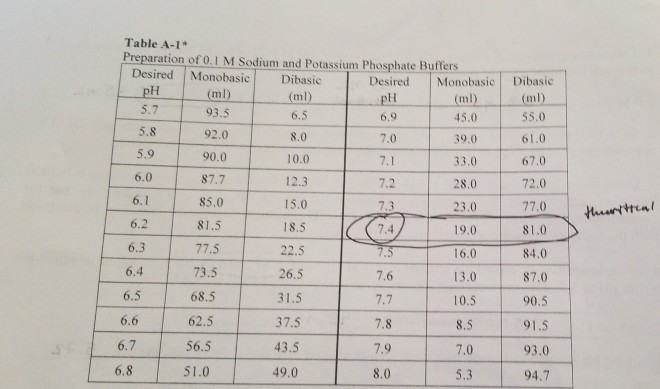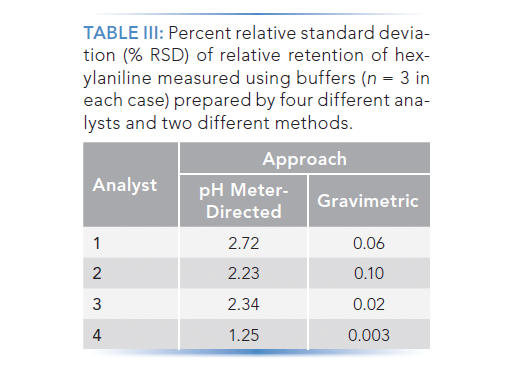
Mobile Phase Buffers in Liquid Chromatography (LC): Effect of Buffer Preparation Method on Retention Repeatability

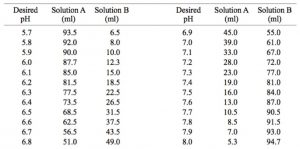
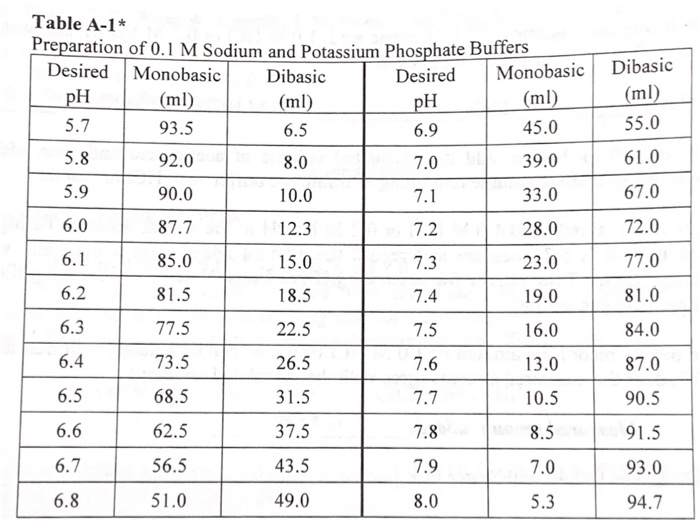
SOLVED: The available matcrials are: M NaOH MHCL KHPO4 (potassium phosphate, monobasic, solid), KHPO4 (potassium phosphate, dibasic, solid) Question -1 (7 pt), If we want t0 prepare 50 mL; pH 7.0, 0.1

SOLVED: A phosphate buffer solution is prepared by dissolving 25.0 g of potassium hydrogen phosphate (K,HPO ; FM = 174.18) and 12.0 g of potassium dihydrogen phosphate (KH,PO : FM = 136.09)