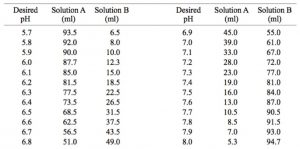
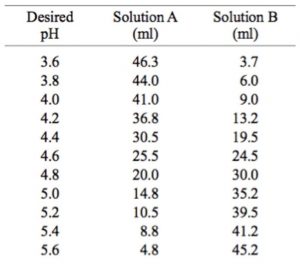
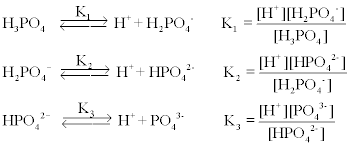SOLVED: A phosphate buffer solution is prepared by dissolving 25.0 g of potassium hydrogen phosphate (K,HPO ; FM = 174.18) and 12.0 g of potassium dihydrogen phosphate (KH,PO : FM = 136.09)

Table 2 from Dynamic approach to predict pH profiles of biologically relevant buffers | Semantic Scholar
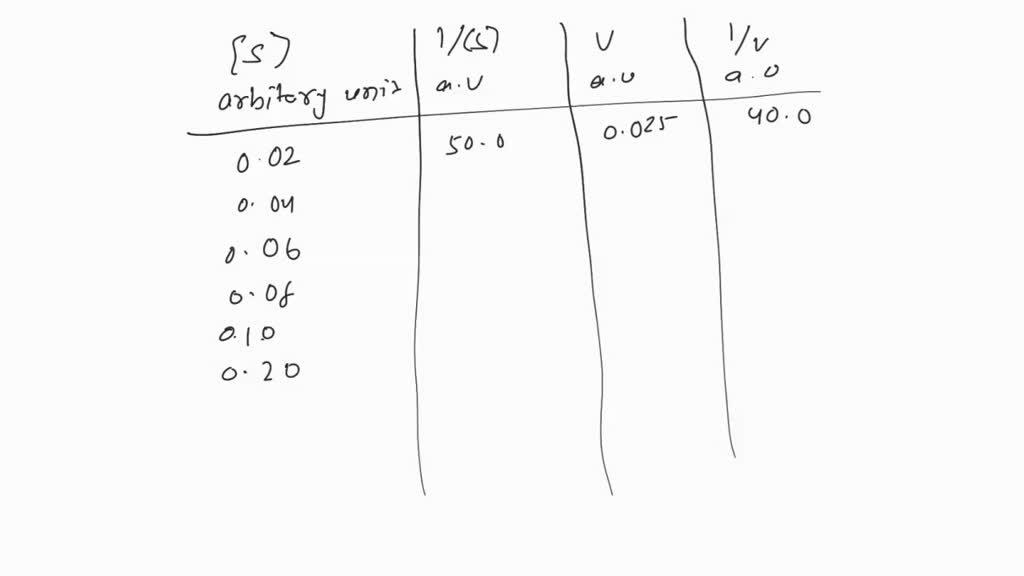
SOLVED: 1.Prepare 100 µL of 0 unit/ml and 0.5 unit/ml β-gal solution in potassium phosphate buffer. Calculate how much stock enzyme is required to prepare the diluted enzyme solution. 2.Prepare 2 tubes