
Calculating Hydronium ion concentration from pH (and identifying molecules based on pH) : r/chemhelp
What is the concentration of hydrogen ion and hydroxide ions of solution which has pH of 4.87? - Quora

How to Calculate ph if Hydrogen ion Concentration is Given For All Stud... | Concentration, Student, Calculator

Calculate the hydrogen ion concentration in the following biological fluids whose pH are given below:(a) Human muscle - fluid, 6.83 (b) Human stomach fluid, 1.2 (c) Human blood, 7.38 (d) Human saliva, 6.4.

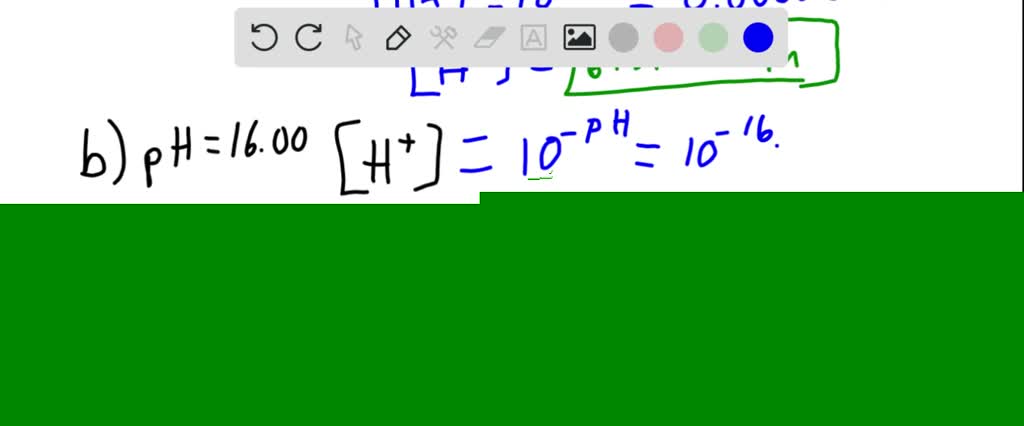
SOLVED:Calculate the hydrogen ion concentration in mol/L for each of the following solutions: (a) a solution whose pH is 5.20, (b) a solution whose pH is 16.00,(c) a solution whose hydroxide concentration


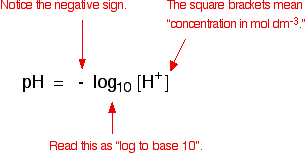

:max_bytes(150000):strip_icc()/how-to-calculate-ph-quick-review-606089_final-165915b0177b4f6e82843f25097f51df.png)






![Calculate [H+] from pH - YouTube Calculate [H+] from pH - YouTube](https://i.ytimg.com/vi/yE4M2Uuh-h4/sddefault.jpg)
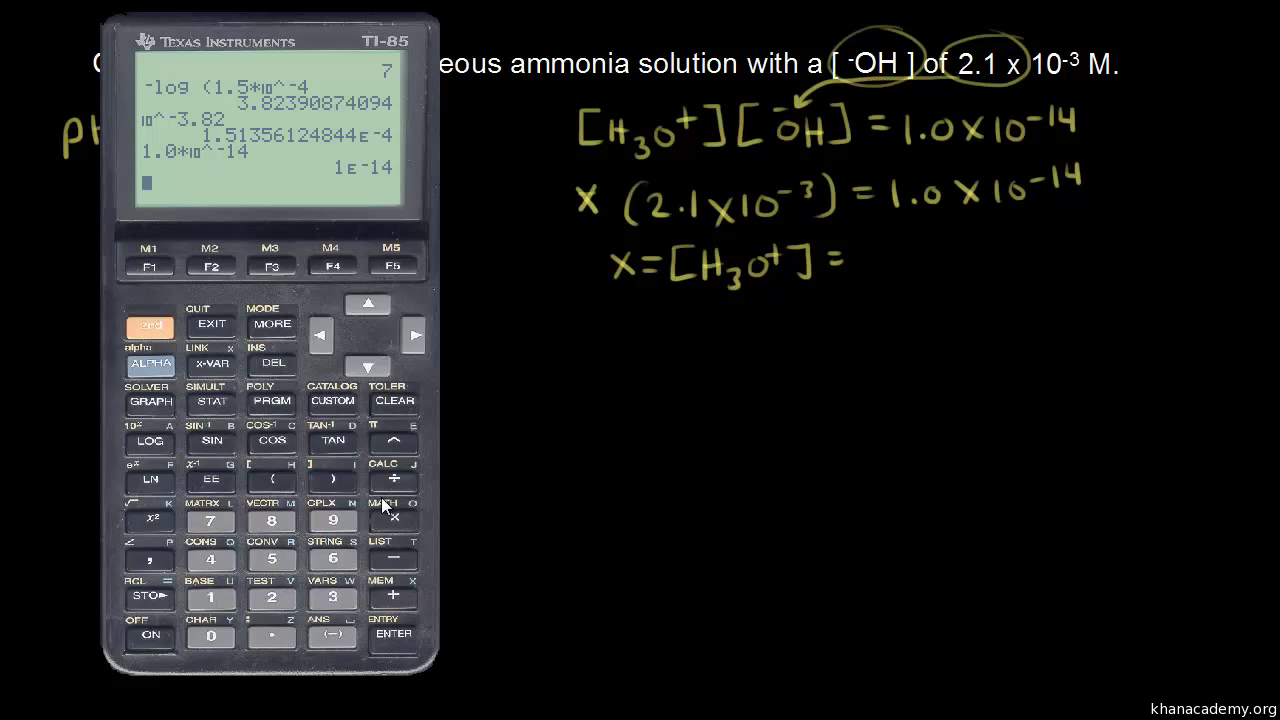


![Calculating [H+] from pH, Acids & Bases Tutorial - YouTube Calculating [H+] from pH, Acids & Bases Tutorial - YouTube](https://i.ytimg.com/vi/bP-evPgNNUg/maxresdefault.jpg)