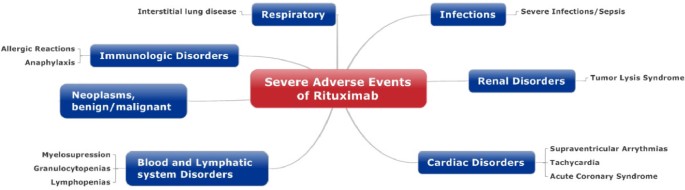
Clinical review: Serious adverse events associated with the use of rituximab - a critical care perspective | Critical Care | Full Text

Rituximab versus intravenous cyclophosphamide in patients with connective tissue disease-associated interstitial lung disease in the UK (RECITAL): a double-blind, double-dummy, randomised, controlled, phase 2b trial - The Lancet Respiratory Medicine

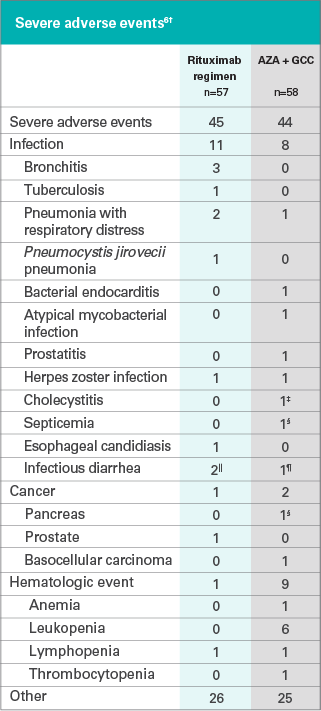
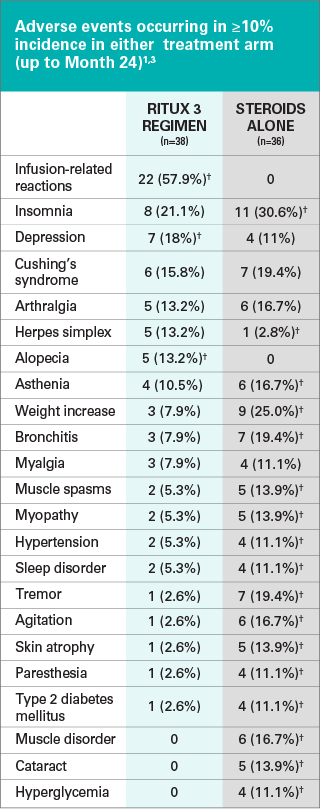
First-line rituximab combined with short-term prednisone versus prednisone alone for the treatment of pemphigus (Ritux 3): a prospective, multicentre, parallel-group, open-label randomised trial - The Lancet

Effect of Immunosuppressive Diseases and Rituximab Infusions on Allowing COVID-19 Infection to Relapse | The Permanente Journal
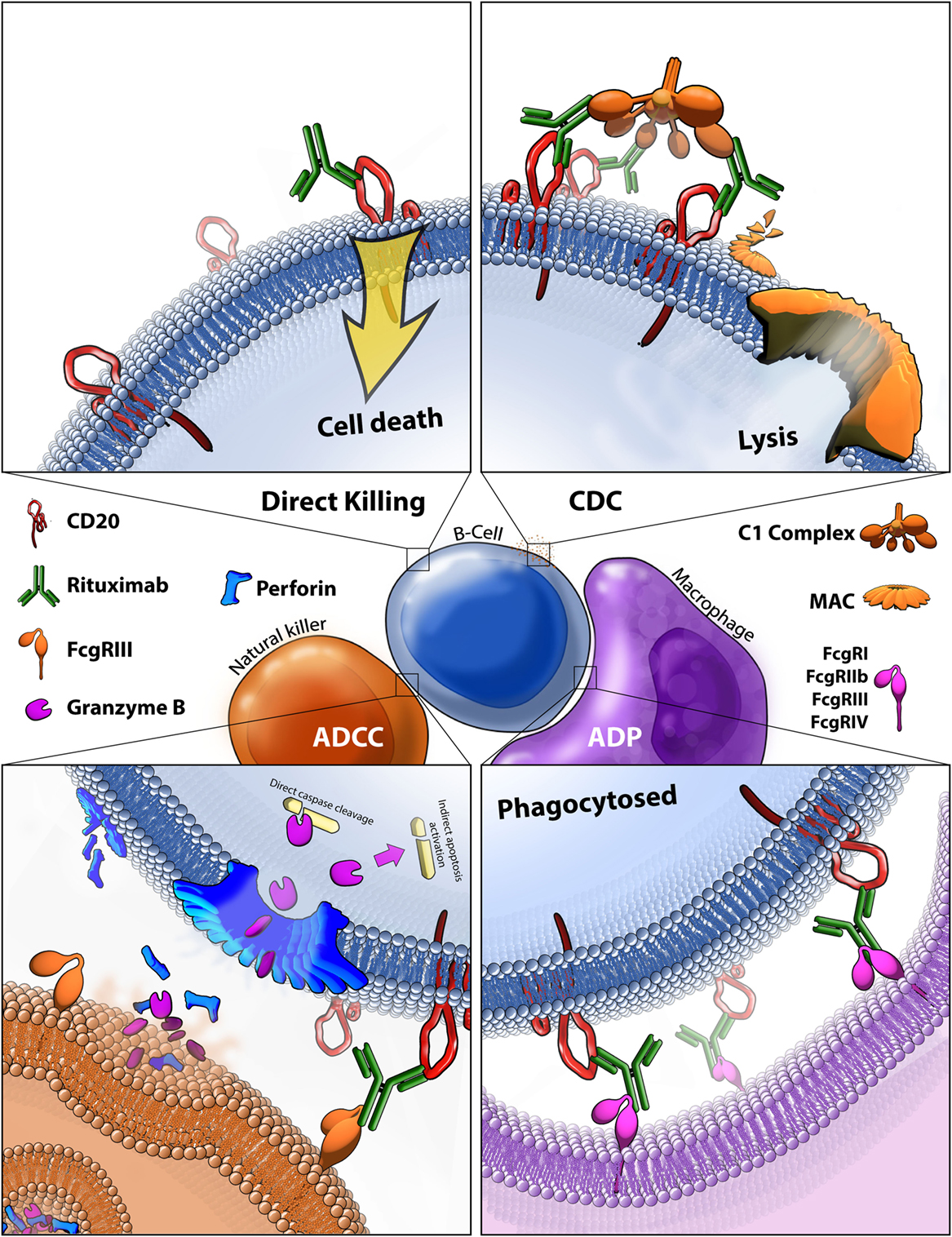
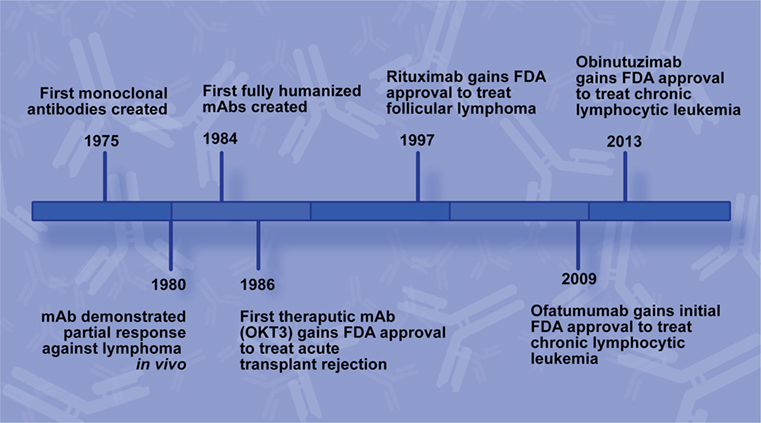
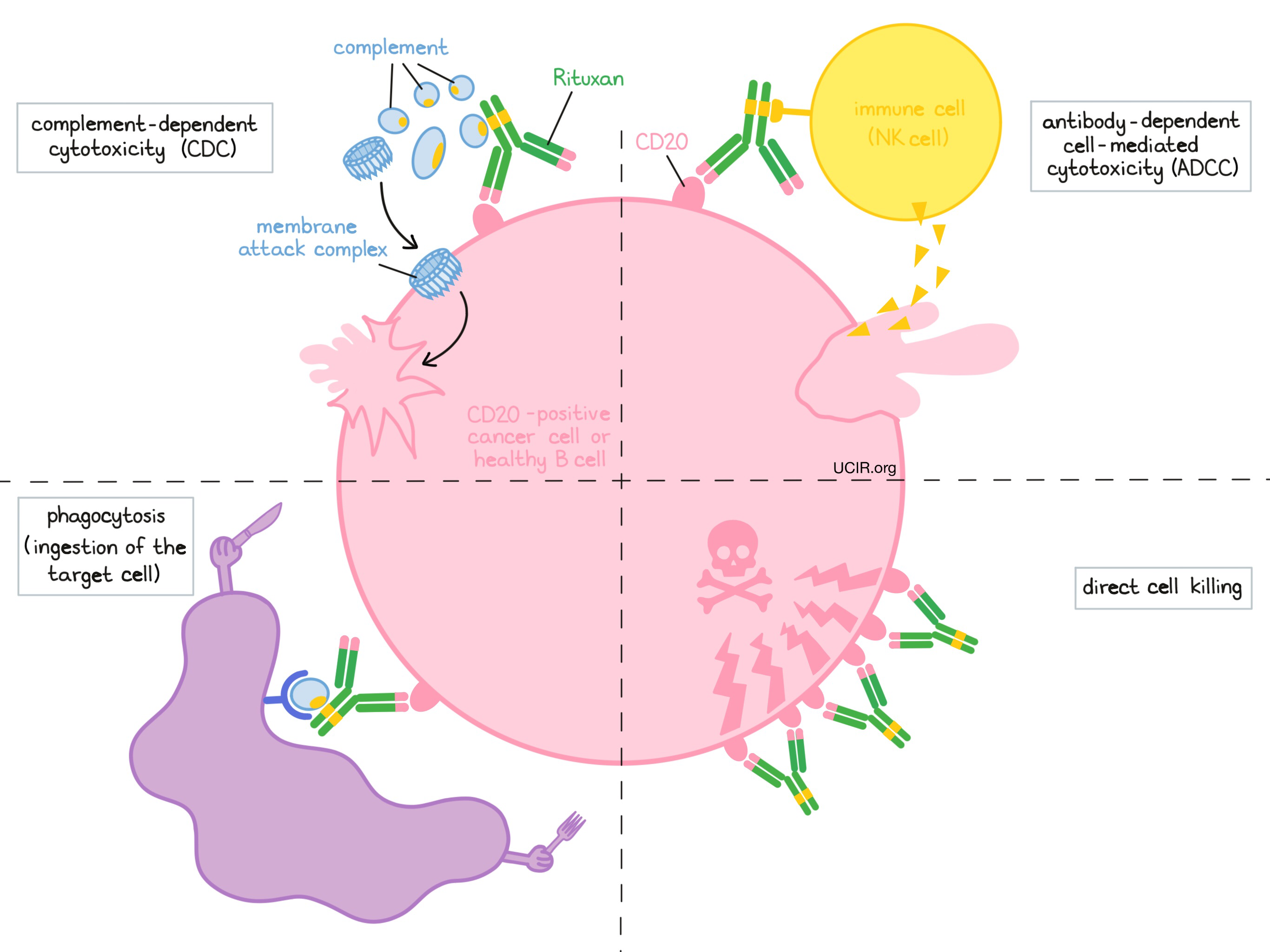
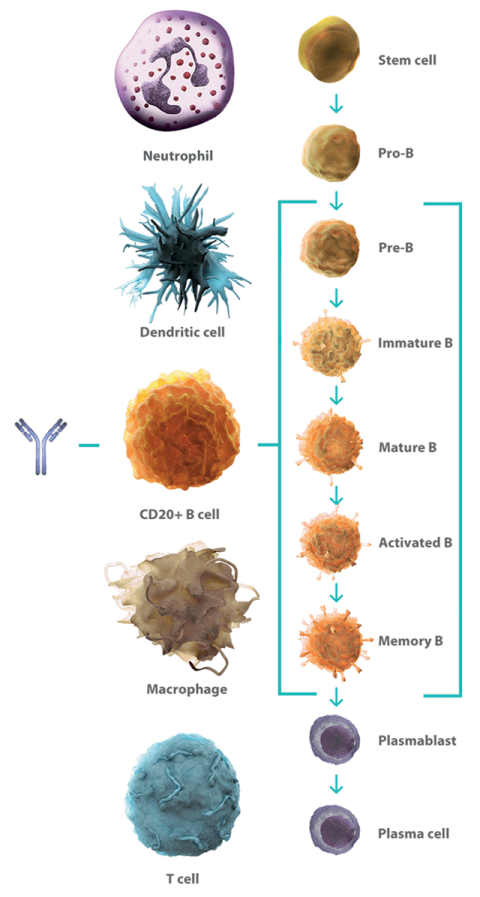
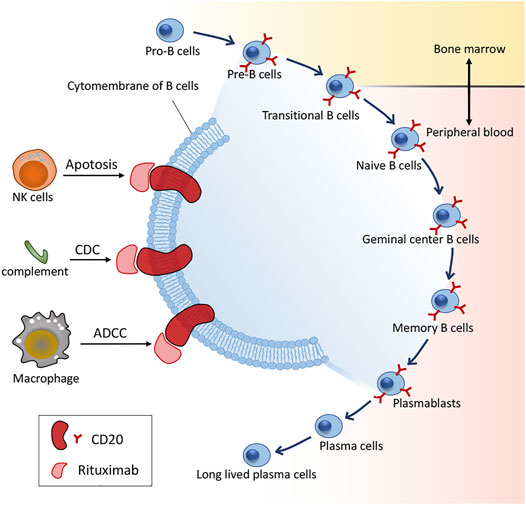
Frontiers | Past, Present, and Future of Rituximab—The World's First Oncology Monoclonal Antibody Therapy