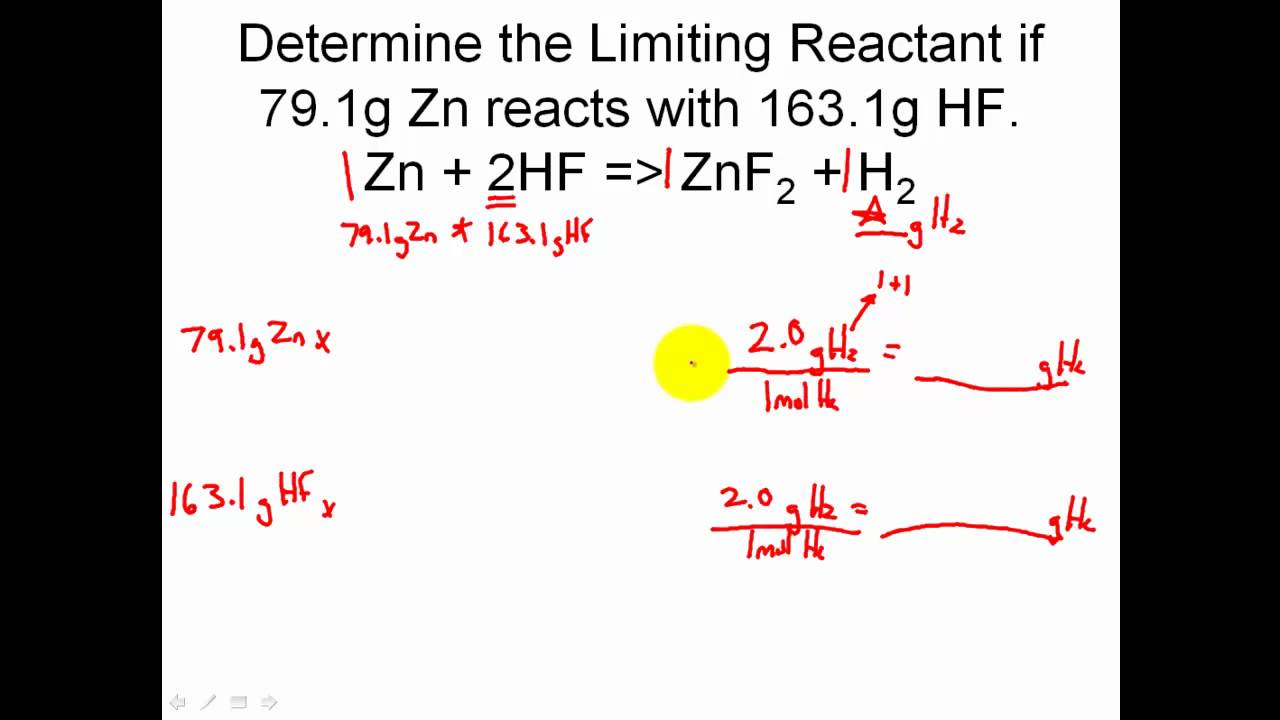
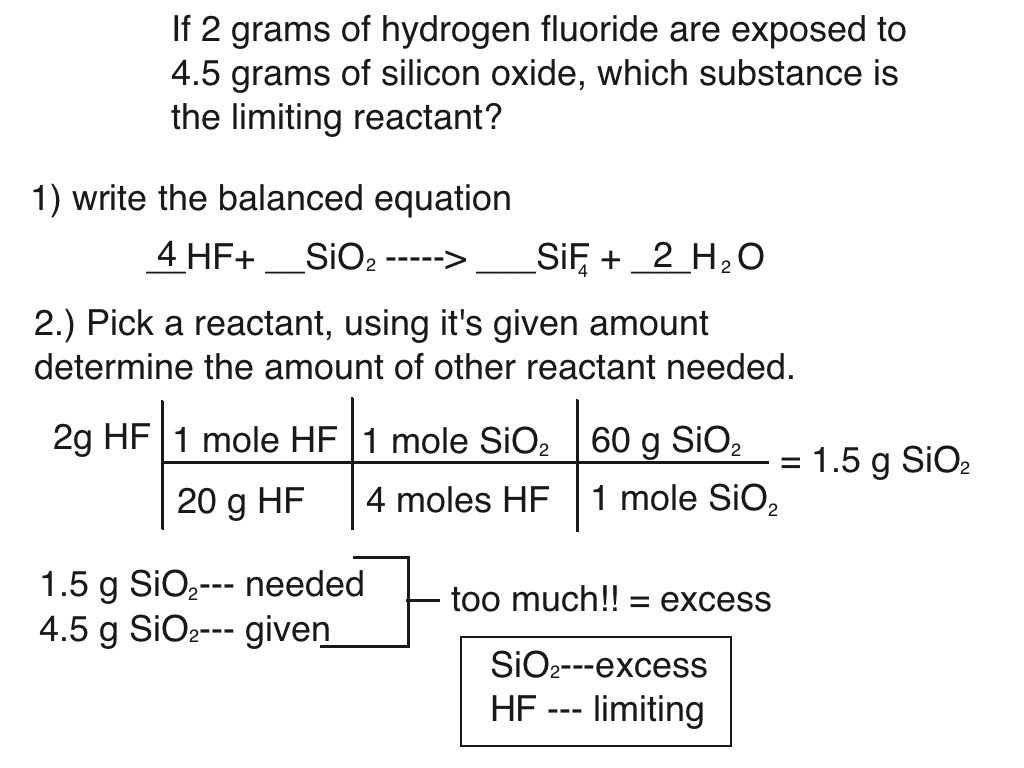
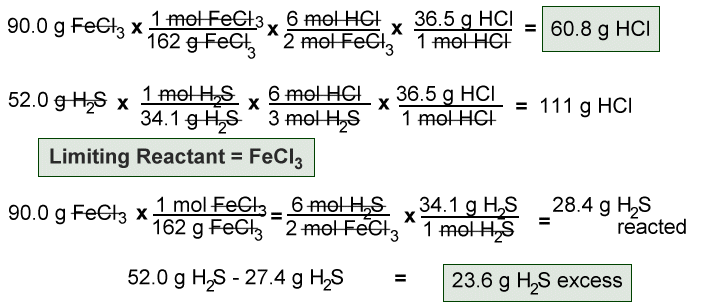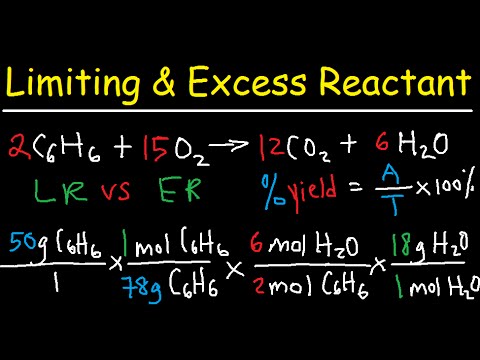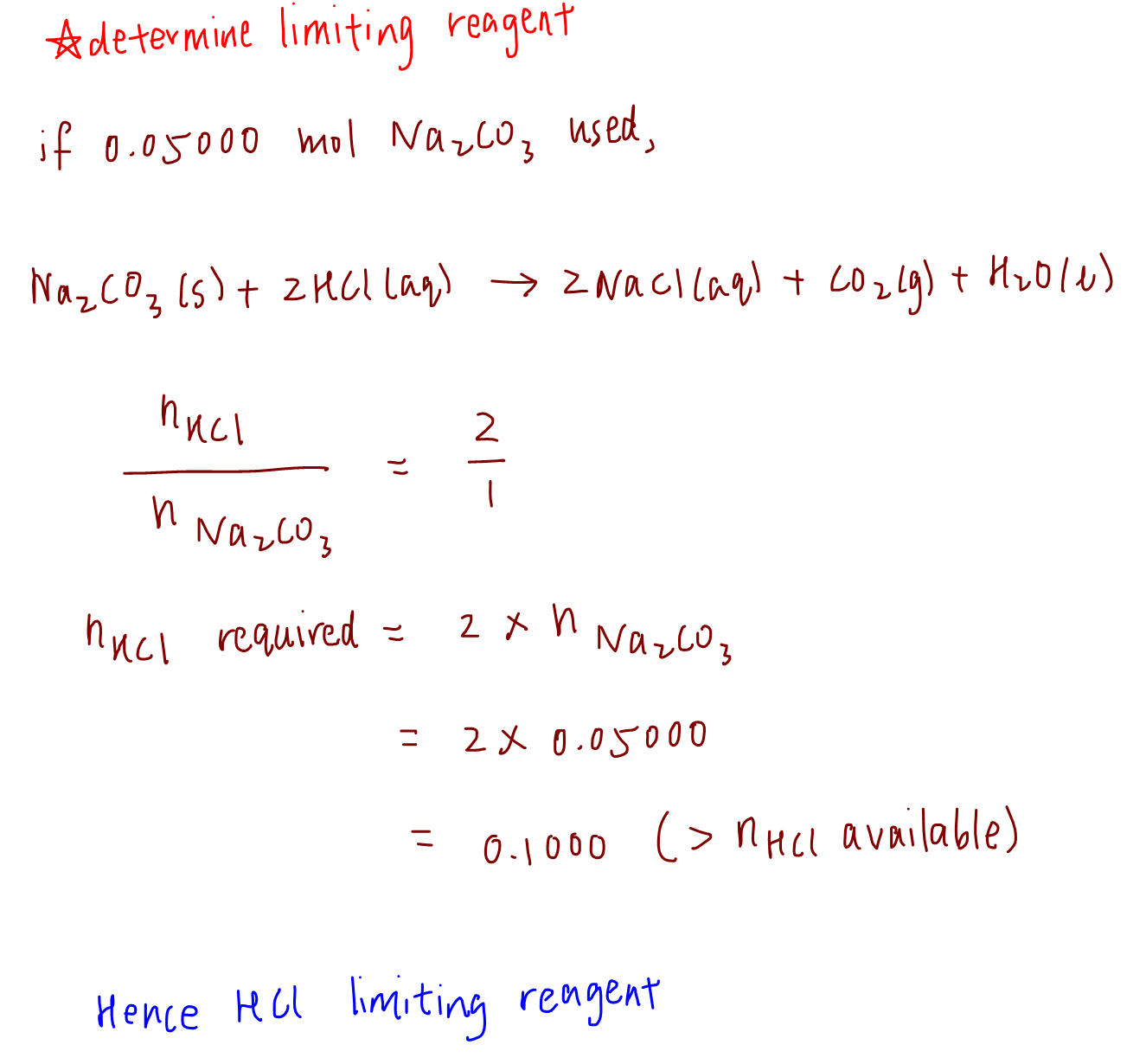How to find Excess and Limiting reactants |Reagents | Chemistry - Best Online Free Chemistry Learning
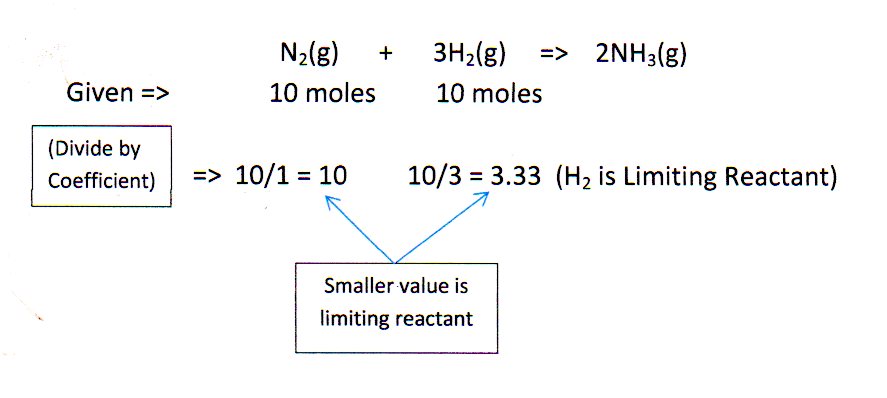
N_2 + 3H_2 -> 2NH_3. What is the limiting reactant if you start the reaction with 10.0 moles of each reactant? | Socratic

CHEM 101: Dimensional Analysis Limiting Reagent, Theoretical Yield, Percent Yield, Excess Reactant 2 - YouTube

How to Find Limiting Reactant (Quick & Easy) Examples, Practice Problems, Practice Questions - YouTube