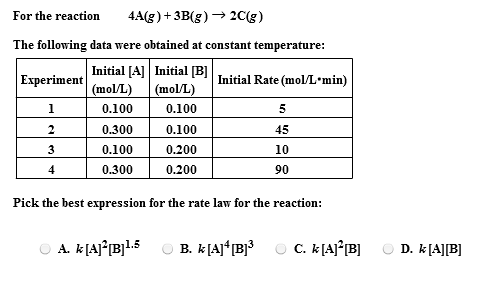
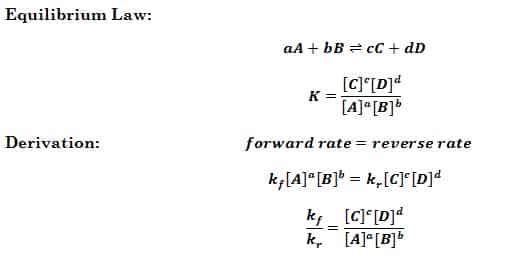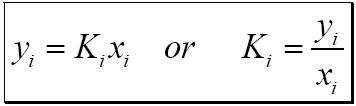Calculate the equation constant for the reaction: H2 (g) + CO2 (g) H2O (g) + CO at 1395 K, if the equilibrium constants at 1395 K for the following are 2H2O (g)

How to Calculate Equilibrium Constant K Value Practice Problems & Exampled Explained Step by Step - YouTube

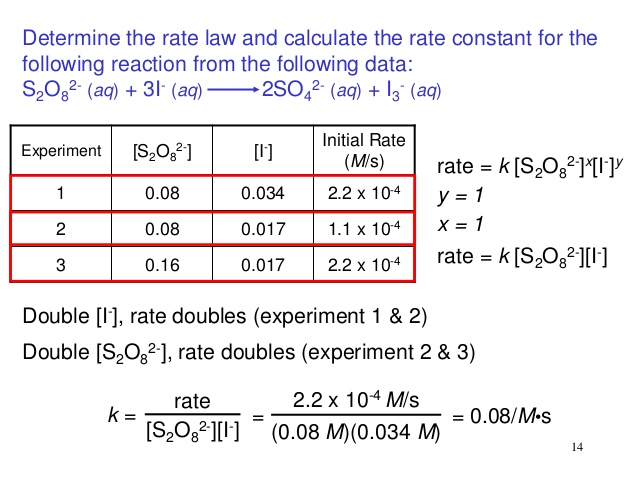
6-3:Calculating Equilibrium Constants: The actual value of K eq is found experimentally. The individual concentrations of all the reactants is calculated, - ppt download
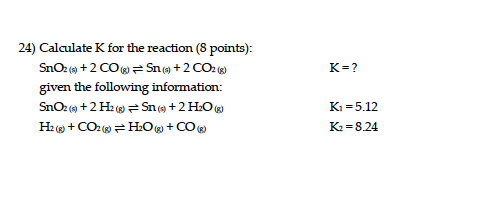





![The constant K used to calculate corrosion rate in eq. 1 [15] | Download Scientific Diagram The constant K used to calculate corrosion rate in eq. 1 [15] | Download Scientific Diagram](https://www.researchgate.net/publication/352980107/figure/tbl2/AS:1041875033145347@1625413773912/The-constant-K-used-to-calculate-corrosion-rate-in-eq-1-15.png)