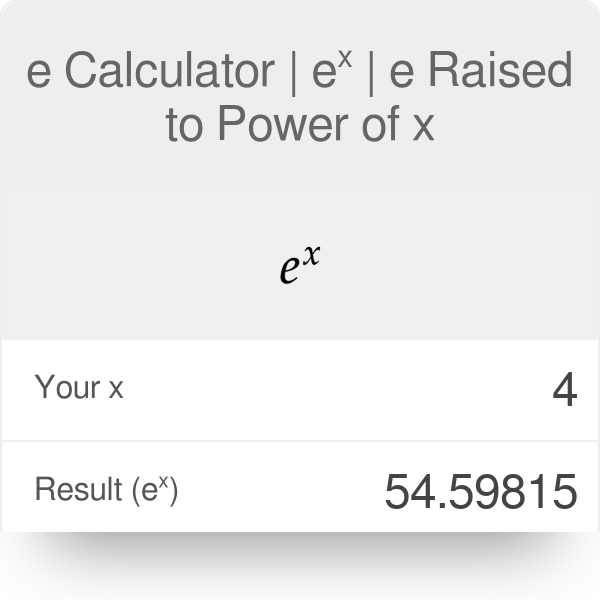
ELECTROCHEMISTRY Chap 20. Electrochemistry Sample Exercise 20.6 Calculating E° cell from E° red Using the standard reduction potentials listed in Table. - ppt download

How to Calculate Standard Cell Potential and Voltage using E cell = E cathode - E anode Examples - YouTube

probability theory - How to calculate $\mathbb{E}\left(T_{n}\right)$ and $\mathbb{E}\left(T^2_{n}\right)$? - Mathematics Stack Exchange

SOLVED: Calculate the sodium chloride equivalent (the E-value) for a substance (use two decimal places) that has a molecular weight (MW) of 136, is a 2 ion electrolyte (and dissociates 80%). The

SOLVED: Calculate E for the following electrochemical cell at 25C Zn(s)| Zn2+ (aq) (0.100M)|| Cu2+ (aq) (0.0750M)| Cu(s) Cu2+ 2e-> Cu(s) E = 0.339 V Zn 2+ (aq) + 2e-> Zn(s) E = -

Methodology used to calculate the E/e ′ ratio in the study population.... | Download Scientific Diagram

calculate e cell and delta g for the following at 28 degrees celcius mg+sn(2+)(0 04M) GIVES MG(2+)(0 06)+sn e - Chemistry - - 9003281 | Meritnation.com

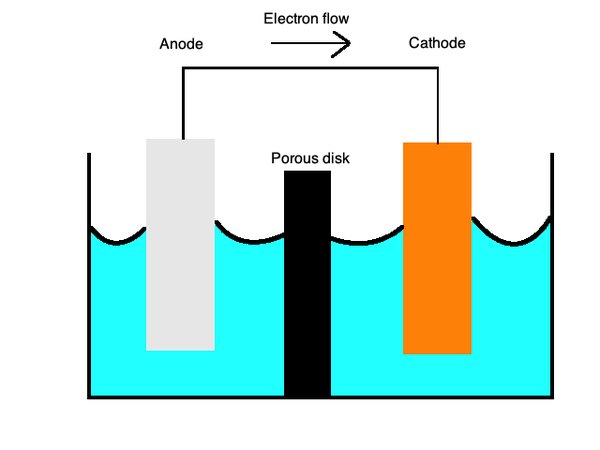
Half-cell potentials Electrochemical Series using E cell predicting reaction feasibility A level GCE AS A2 chemistry revision notes KS5
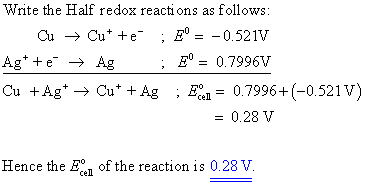


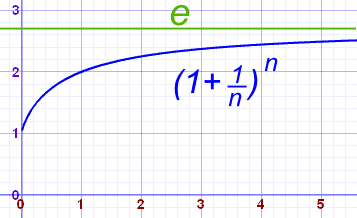



![probability - How to calculate $E[X]$ of a Poisson random variable. - Mathematics Stack Exchange probability - How to calculate $E[X]$ of a Poisson random variable. - Mathematics Stack Exchange](https://i.stack.imgur.com/utxeE.png)