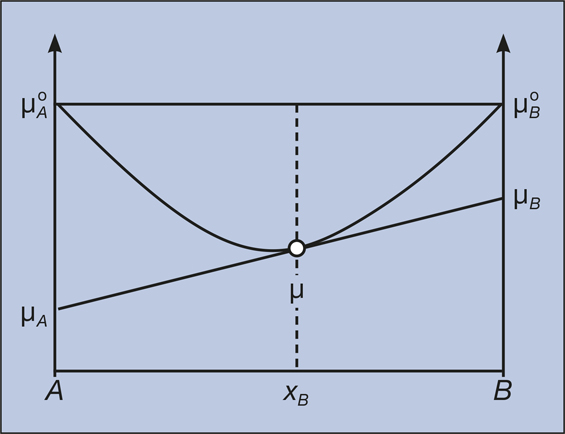
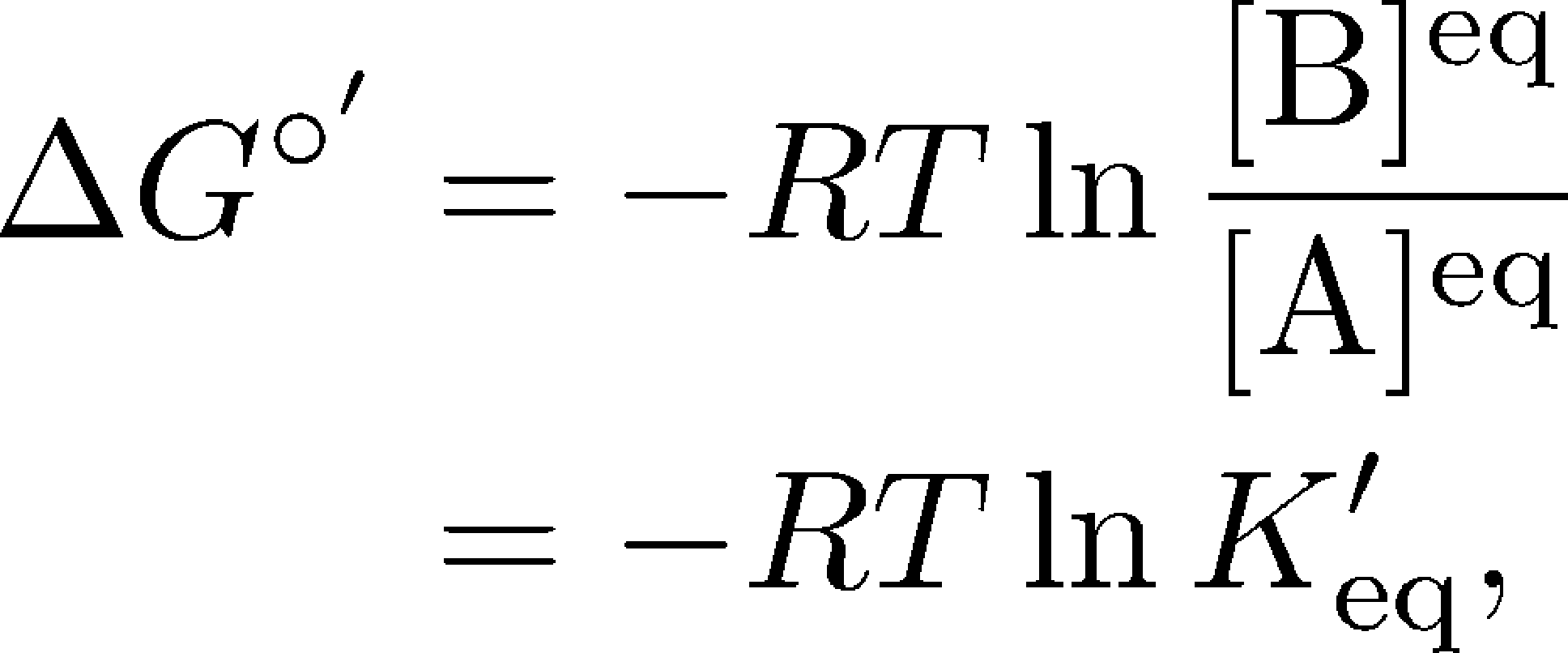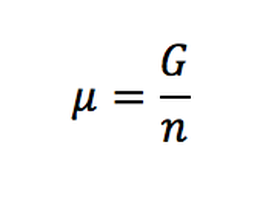What is the driving force for mass transfer, chemical potential or concentration difference and what is exactly chemical potential? - Quora
![SOLVED: Calculate the chemical potential difference When Na" mM; extracellular [Na ] transported into the cells at 379C . Intracellular [Na ] R-8.314 J*K 150 mM; the membrane potential mol" 10 pts SOLVED: Calculate the chemical potential difference When Na" mM; extracellular [Na ] transported into the cells at 379C . Intracellular [Na ] R-8.314 J*K 150 mM; the membrane potential mol" 10 pts](https://cdn.numerade.com/ask_images/543d027fef634e99a2292ef79b8dbe8d.jpg)
SOLVED: Calculate the chemical potential difference When Na" mM; extracellular [Na ] transported into the cells at 379C . Intracellular [Na ] R-8.314 J*K 150 mM; the membrane potential mol" 10 pts
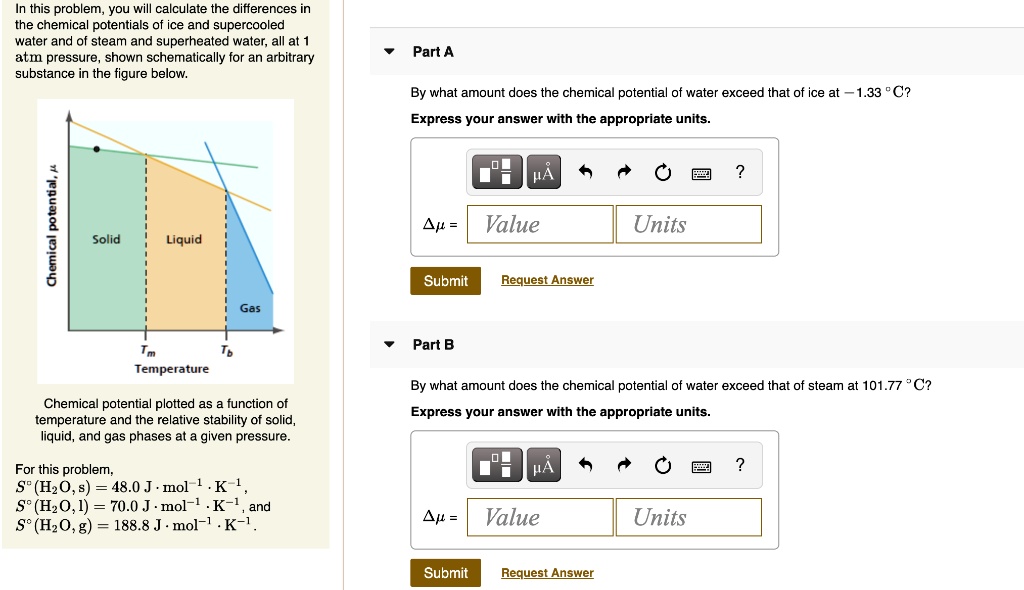
SOLVED: this problem, you calculate the differences in the chemical potentials of ice and supercooled water and of steam and superheated water; all at 1 atmn pressure, shown schematically for an arbitrary
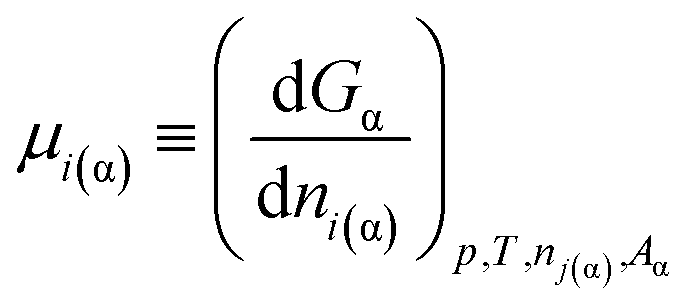
A new paradigm on the chemical potentials of components in multi-component nano-phases within multi-phase systems - RSC Advances (RSC Publishing) DOI:10.1039/C7RA07911G

SOLVED: The enthalpy of a pure substance is given by the equation 652 P H = In Po where and Po are constants, S is the entropy; P is the pressure, and