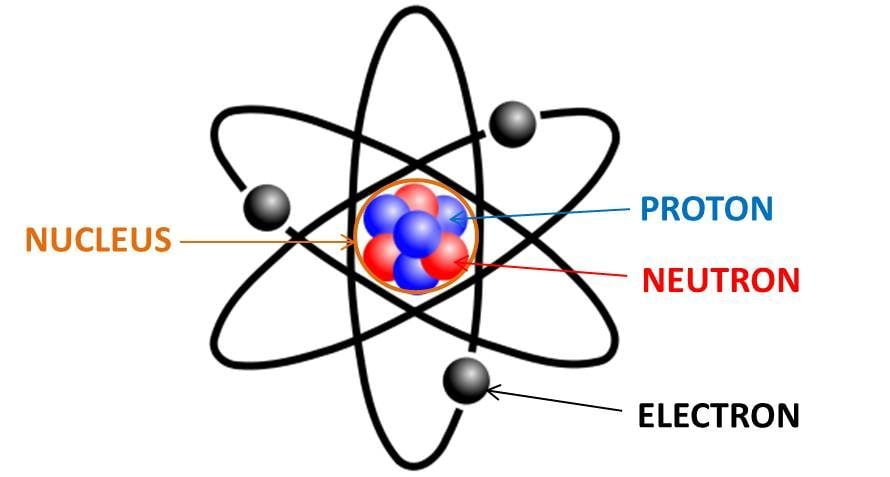
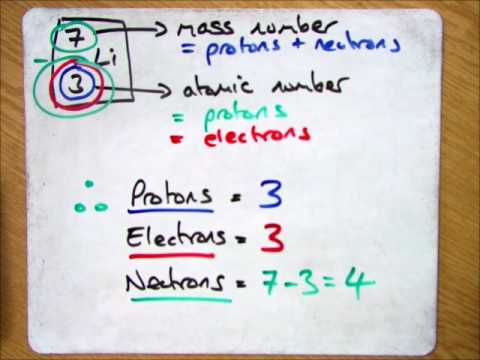
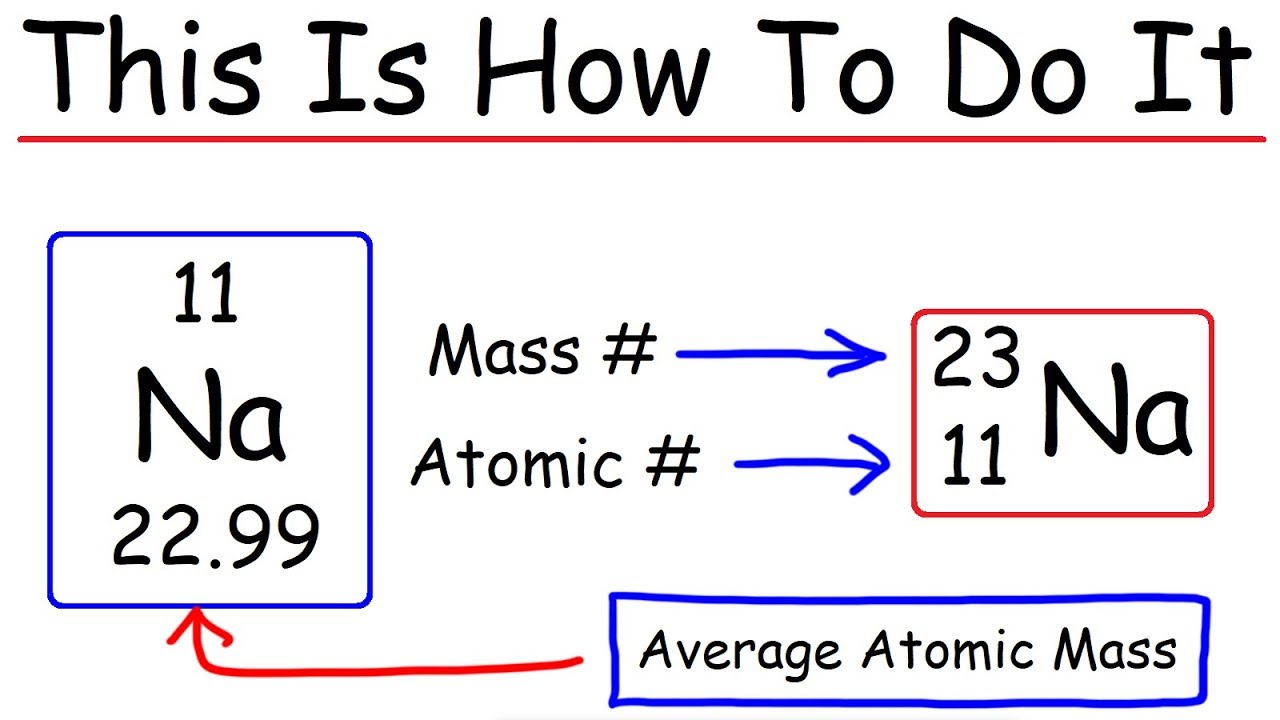
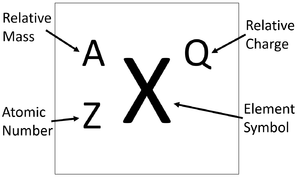
2. Calculate the number of neutrons in the following atoms : a) 13A1 27 b) ispl3. Make a model of - Brainly.in
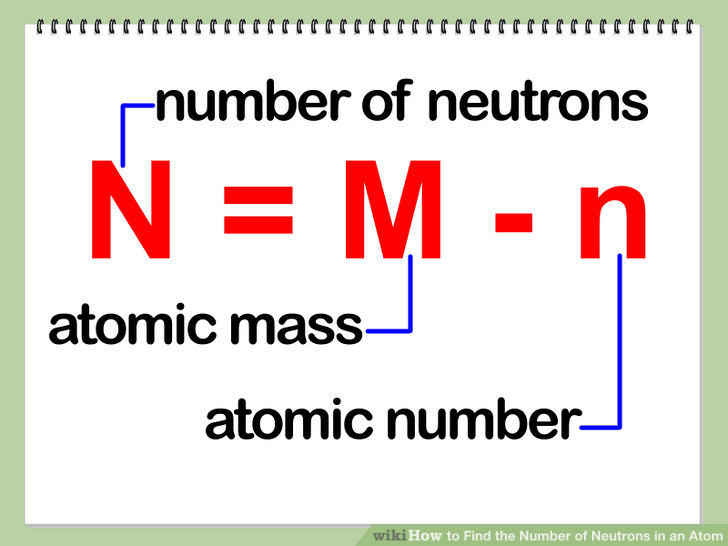
The mass number of a chromium atom is 52 and it has 24 protons. How many neutrons does this atom have? | Socratic
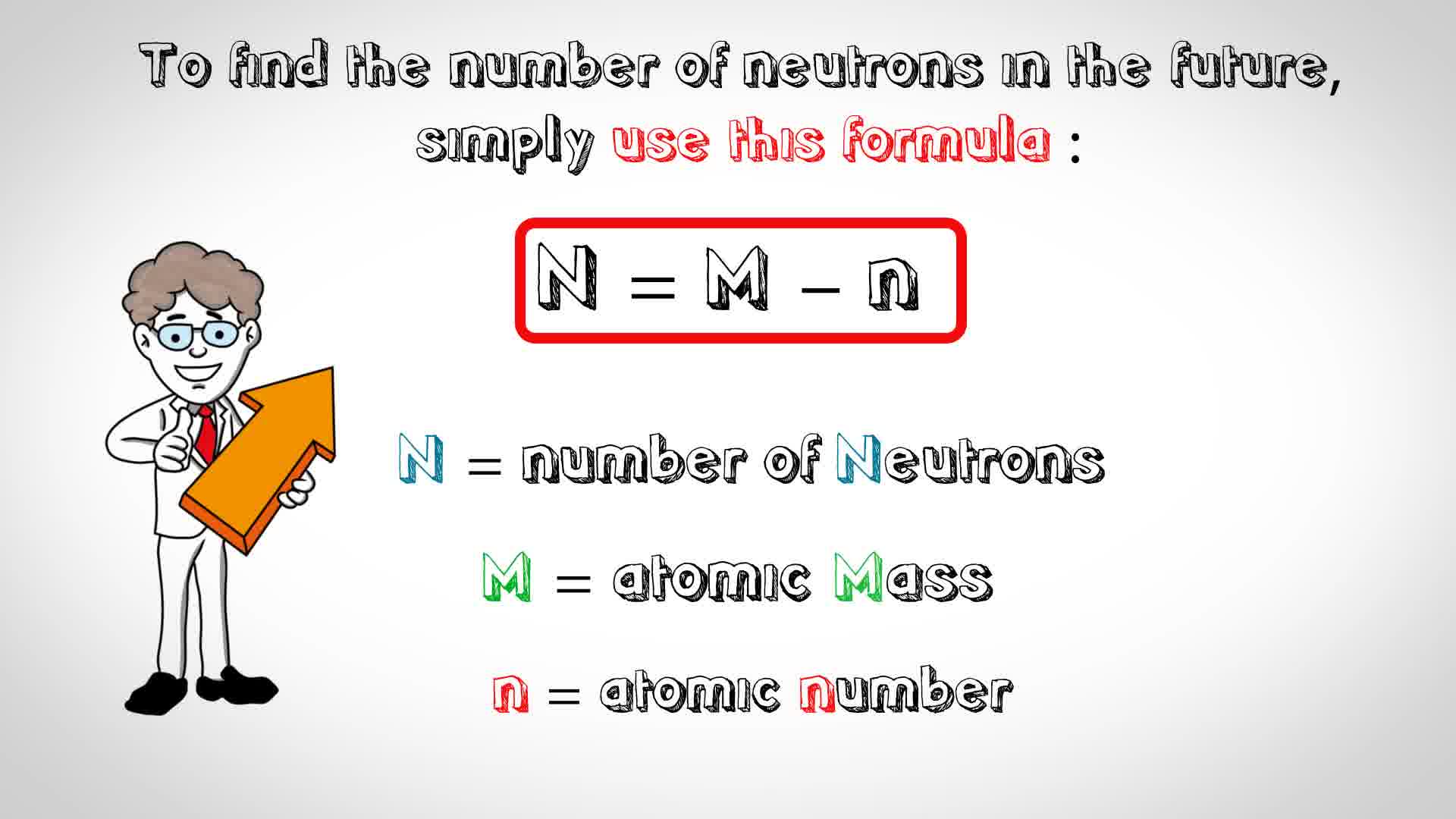
Zirconium has an atomic number of 40. How many neutrons are there in an isotope of zirconium-92? | Socratic

An element X has 7 electrons in its L shell. What is true about the element X ?I. It belongs to period 9 of modern periodic table.II. Its atom contains 9 protons.III.
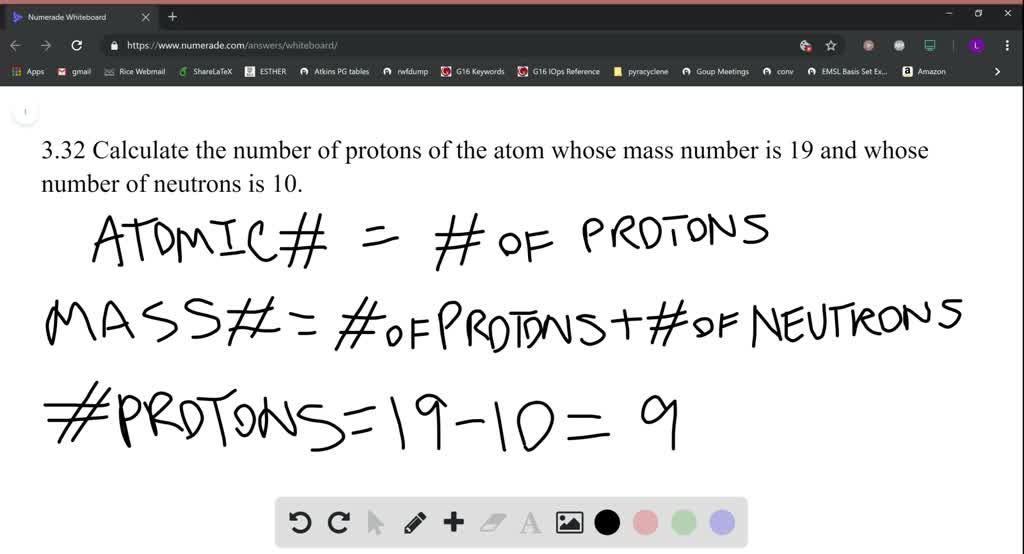
SOLVED:Calculate the number of protons of the atom whose mass number is 19 and whose number of neutrons is 10.